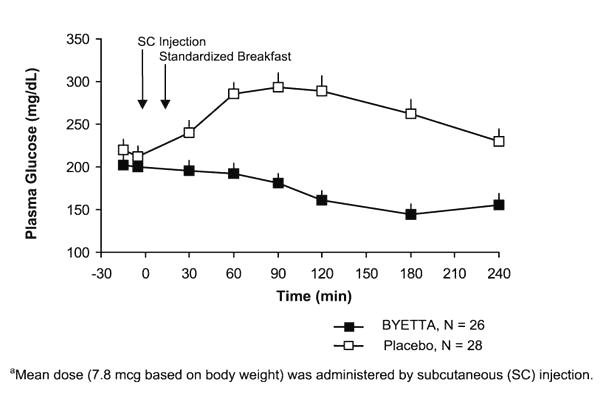
They are also known as incretin mimetics. It is an injectable drug that reduces blood glucose by increasing insulin production by the pancreas, slows the absorption of glucose from the gut, reduces the activity of glucagon (a hormone that increases release of glucose from the liver), and reduces appetite. What does Byetta treat. Type 2 diabetes mellitus in conjunction with diet and exercise. It is not used for treating type- 1 diabetes or diabetic ketoacidosis and it should not be used in place of insulin when patients require insulin. How effective is Byetta. Monotherapy: Byetta 5 to 1. Hb. A1c from baseline better than placebo. The average reductions in Hb. A1c were - 0. 2% for placebo, - 0. Byetta 5 mcg, and - 0. Byetta 1. 0 mcg. Combination therapy: Byetta 5 to 1. Hb. A1c from baseline better than placebo after 3. Changes in Hb. A1c were - 0. Byetta 5 mcg/metformin, and - 0. Byetta 1. 0 mcg/metformin; +0. Byetta 5 mcg/sulfonylurea, - 0. Byetta 1. 0 mcg/sulfonylurea; +0. Byetta 1. 0 mcg/pioglitazone or rosiglitazone. Byetta versus Victoza: In one 2. Victoza 1. 8 mg daily reduced Hb. A1c more than Byetta 1. Victoza achieved Hb. A1c below 7%. Average reduction in weight was similar between both drugs (- 3. What are interesting facts about Byetta. First FDA approved incretin mimetic. Patients can develop antibodies which may reduce the effect of Byetta. Learn about BYETTA, the BYETTA Pen. Cases are being reviewed nationwide for injuries caused as a result of the following dangerous medication side effects. Find a comprehensive guide to possible side effects including common and rare side effects when taking Byetta (Exenatide Injection) for healthcare professionals and. Byetta 5 micrograms solution for injection, prefilled pen. Byetta 10 micrograms solution for injection, prefilled pen. Medscape - Type 2 diabetes-specific dosing for Byetta (exenatide), frequency-based adverse effects, comprehensive interactions, contraindications, pregnancy. Credit: Thinkstock Most, if not all, the studies of mindful eating and drinking have focused on weight loss rather than on diabetes. But losing weight matters to.
 
Byetta may reduce appetite and cause weight loss. There is a once- weekly extended- release formulation of exenatide called Bydureon. What are the side effects of Byetta. Common: nausea, vomiting, diarrhea, nervousness, dizziness, increased sweating, headache, acid reflux, reduced appetite, stomach discomfort, and injection site reactions. The risk of hypoglycemia (low blood glucose) is increased when combined with a sulfonylurea. Serious: Pancreatitis, serious allergic reactions, severely low blood glucose (hypoglycemia), kidney failure. 
What is the dosing of Byetta BYETTA pen is available as pre- filled multiple dose pen: 3. L 6. 00 mcg/2. 4 m. LThe initial recommended dosage of Byetta pen is 5 mcg injected under skin (thigh, abdomen, or upper arm) twice daily 6. After 1 month, the dosage can be increased to Byetta 1. Byetta drug interactions. Warfarin (Coumadin, Jantoven): Byetta may increase the effect of warfarin, leading to more bleeding. Byetta slows movement of food and drugs through the intestines and therefore can reduce the absorption of oral drugs. Oral medications should be administered one hour before injecting Byetta. What is the price of Byetta. Byetta costs $2. 00 to $3. How to obtain prescription cost assistance for Byetta. For prescription assistance visit: My. Savings. Rx Card. BMS Patient Assistance Program. Reference: Byetta FDA Prescribing Information and medication Guide. Comment on this article or post an article by joining the Rx. Econsult community. Please Share on Your Social Networks. This medication summary is for information only and is not a substitute for medical advice, diagnosis or treatment provided by a qualified health care provider. Official BYETTA. After initiation and dose increases of BYETTA, observe patients carefully for pancreatitis (including persistent severe abdominal pain, sometimes radiating to the back, with or without vomiting). If pancreatitis is suspected, BYETTA should be discontinued promptly and should not be restarted if pancreatitis is confirmed. Hypoglycemia: Increased risk of hypoglycemia when used in combination with a sulfonylurea (SU) or when used with a glucose- independent insulin secretagogues (eg, meglitinides). Clinicians may consider reducing the SU dose in patients receiving BYETTA to reduce the risk of hypoglycemia. When used with insulin, evaluate and consider reducing the insulin dose in patients at increased risk of hypoglycemia. Renal Impairment: Should not be used in patients with severe renal impairment or end- stage renal disease. Use with caution in patients with renal transplantation or when initiating or escalating the dose in patients with moderate renal failure. Postmarketing reports of altered renal function, including increased serum creatinine, renal impairment, worsened chronic renal failure, and acute renal failure, sometimes requiring hemodialysis and kidney transplantation. Gastrointestinal Disease: Because exenatide is commonly associated with gastrointestinal adverse reactions, BYETTA is not recommended in patients with severe gastrointestinal disease (eg gastroparesis). Immunogenicity: Patients may develop antibodies to exenatide. In 3 registration trials, antibody levels were measured in 9. If worsening of or failure to achieve adequate glycemic control occurs, consider alternative antidiabetic therapy. Hypersensitivity: Postmarketing reports of serious hypersensitivity reactions (eg anaphylaxis and angioedema). If this occurs, patients should discontinue BYETTA and other suspect medications and promptly seek medical advice. Macrovascular Outcomes: No clinical studies establishing conclusive evidence of macrovascular risk reduction with BYETTA or any other antidiabetic drug. Most Common Adverse Reactions (=5%)2. PBO): nausea (8% vs 0%). Three 3. 0- week combination trials of BYETTA added to metformin (MET) and/or SU vs PBO: nausea (4. BYETTA added to thiazolidinedione (TZD) . Three 3. 0- week combination trials of BYETTA added to MET and/or SU vs PBO: nausea (3% vs < 1%), vomiting (1% vs 0). BYETTA added to TZD . BYETTA added to insulin glargine . Use with caution with medications that have a narrow therapeutic index or require rapid gastrointestinal absorption. Oral medications dependent on threshold concentrations for efficacy, such as contraceptives or antibiotics, should be taken at least 1 hour before BYETTA. Warfarin: Postmarketing reports of increased international normalized ratio (INR) sometimes associated with bleeding with concomitant use of warfarin. Monitor INR frequently until stable upon initiation or alteration of BYETTA. Use in Specific Populations. Pregnant and Nursing Women: Based on animal data, BYETTA may cause fetal harm and should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. To report drug exposure during pregnancy call 1- 8. When administered to a nursing woman, a decision should be made whether to discontinue nursing or discontinue BYETTA. Pediatric Patients: Use in pediatric patients is not recommended as safety and effectiveness have not been established. Indication and Important Limitations of Use for BYETTABYETTA is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. Not a substitute for insulin and should not be used in patients with type 1 diabetes or diabetic ketoacidosis. Concurrent use with prandial insulin has not been studied and cannot be recommended. BYETTA has been associated with acute pancreatitis, including fatal and non- fatal hemorrhagic or necrotizing pancreatitis, based on postmarketing data. It is unknown whether patients with a history of pancreatitis are at increased risk for pancreatitis while using BYETTA; consider other antidiabetic therapies for these patients. Please see full Prescribing Informationand Medication Guidefor BYETTA (exenatide) injection 5 mcg and 1. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www. fda. gov/medwatch or call 1- 8. FDA- 1. 08. 8. The site you are about to visit is maintained by a third party who is solely responsible for its contents. Astra. Zeneca provides this link as a service to website visitors. Astra. Zeneca is not responsible for the privacy policy of any third- party websites. We encourage you to read the privacy policy of every website you visit. Continue. GO BACK References: 1. Buse JB, Bergenstal RM, Glass LC, et al. Use of twice- daily exenatide in basal insulin–treated patients with type 2 diabetes: a randomized, controlled trial. Ann Intern Med. 2. Riddle MC, Rosenstock J, Gerich J; Insulin Glargine 4. Study Investigators. The treat- to- target trial: randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients. Diabetes Care. 2. Reference: 1. Diamant M, Nauck MA, Shaginian R, et al; for the 4. B Study Group. Glucagon- like peptide- 1 receptor agonist or bolus insulin with optimized basal insulin in type 2 diabetes. Diabetes Care. 2. References: 1. Diamant M, Nauck MA, Shaginian R, et al; for the 4. B Study Group. Glucagon- like peptide- 1 receptor agonist or bolus insulin with optimized basal insulin in type 2 diabetes. Diabetes Care. 2. Yki- Jarvinen H, Juurinen L, Alvarsson M, et al. Initiate Insulin by Aggressive Titration and Education (INITIATE): a randomized study to compare initiation of insulin combination therapy in type 2 diabetic patients individually and in groups. Diabetes Care. 2. References: 1. Diamant M, Nauck MA, Shaginian R, et al; for the 4. B Study Group. Glucagon- like peptide- 1 receptor agonist or bolus insulin with optimized basal insulin in type 2 diabetes. Diabetes Care. 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
August 2017
Categories |
 RSS Feed
RSS Feed
